The atmospheric pressure at sea level is 14.7 psi. Atmospheric pressure is also often stated as pounds/square inch (psi).

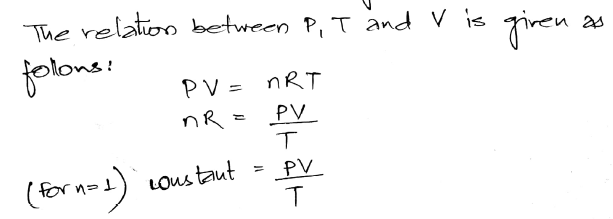
x 104 g/L when the temperature is 15oC and the pressure is 780 mm Hg. Standard atmospheric pressure is called 1 atm of pressure and is equal to 760 mmHg and 101.3 kPa. of 26 g occupies a volume of 392 L at 32oC and at a pressure of 0.95 atm. Another commonly used unit of pressure is the atmosphere (atm).
A pascal is a very small amount of pressure, so the more useful unit for everyday gas pressures is the kilopascal (kPa). The pascal (Pa) is the standard unit of pressure. An equivalent unit to the mmHg is called the torr, in honor of the inventor of the barometer, Evangelista Torricelli. One unit of gas pressure is the millimeter of mercury (mmHg). Tire pressure is best measured when the tire is cold since driving the car for a while will heat up the air in the tire and increase the pressure.Ī barometer measures gas pressure by the height of the column of mercury. The pressure on the tire is the maximum pressure for that tire, not the recommended one.
#780 MMHG TO ATM MANUAL#
The recommended pressure for that model of car (usually somewhere between 32-35 psi) is usually listed in the owner’s manual or stamped somewhere inside the door. The car gets better gas mileage and the tires don’t wear out as fast. The ride is smoother and safer than with lowered pressure.

If you actually perch a bowling ball on your thumbnail, the pressure experienced is twice the usual pressure, and the sensation is unpleasant.There are several benefits to maintaining the proper air pressure in a car tire. These may seem like huge amounts, and they are, but life on earth has evolved under such atmospheric pressure. At sea level, this pressure is roughly the same as that exerted by a full-grown African elephant standing on a doormat, or a typical bowling ball resting on your thumbnail. The atmosphere above us exerts a large pressure on objects at the surface of the earth, roughly equal to the weight of a bowling ball pressing on an area the size of a human thumbnail.Ī dramatic illustration of atmospheric pressure is provided in this brief video, which shows a railway tanker car imploding when its internal pressure is decreased.Ī smaller scale demonstration of this phenomenon is briefly explained.Ītmospheric pressure is caused by the weight of the column of air molecules in the atmosphere above an object, such as the tanker car. In fact, normal air pressure is strong enough to crush a metal container when not balanced by equal pressure from inside the container. Although the force of each collision is very small, any surface of appreciable area experiences a large number of collisions in a short time, which can result in a high pressure. Gas pressure is caused by the force exerted by gas molecules colliding with the surfaces of objects ( Figure 1). Although we do not normally notice atmospheric pressure, we are sensitive to pressure changes-for example, when your ears “pop” during take-off and landing while flying, or when you dive underwater. The earth’s atmosphere exerts a pressure, as does any other gas.

By the end of this section, you will be able to: At a pressure of 780.0 mm Hg and 24.2 C, a certain gas has a volume of 350.0 mL.
